Galderma Neuromodulators

Relfydess ™
Relfydess™ ▼* (RelabotulinumtoxinA) is the first and only ready-to-use liquid neuromodulator created with PEARL™ Technology, developed and manufactured by Galderma.
.jpg)
Azzalure (Botulinum Toxin Type A)
Approved for temporary improvement in the appearance of glabellar and lateral canthal lines in adults under 65.
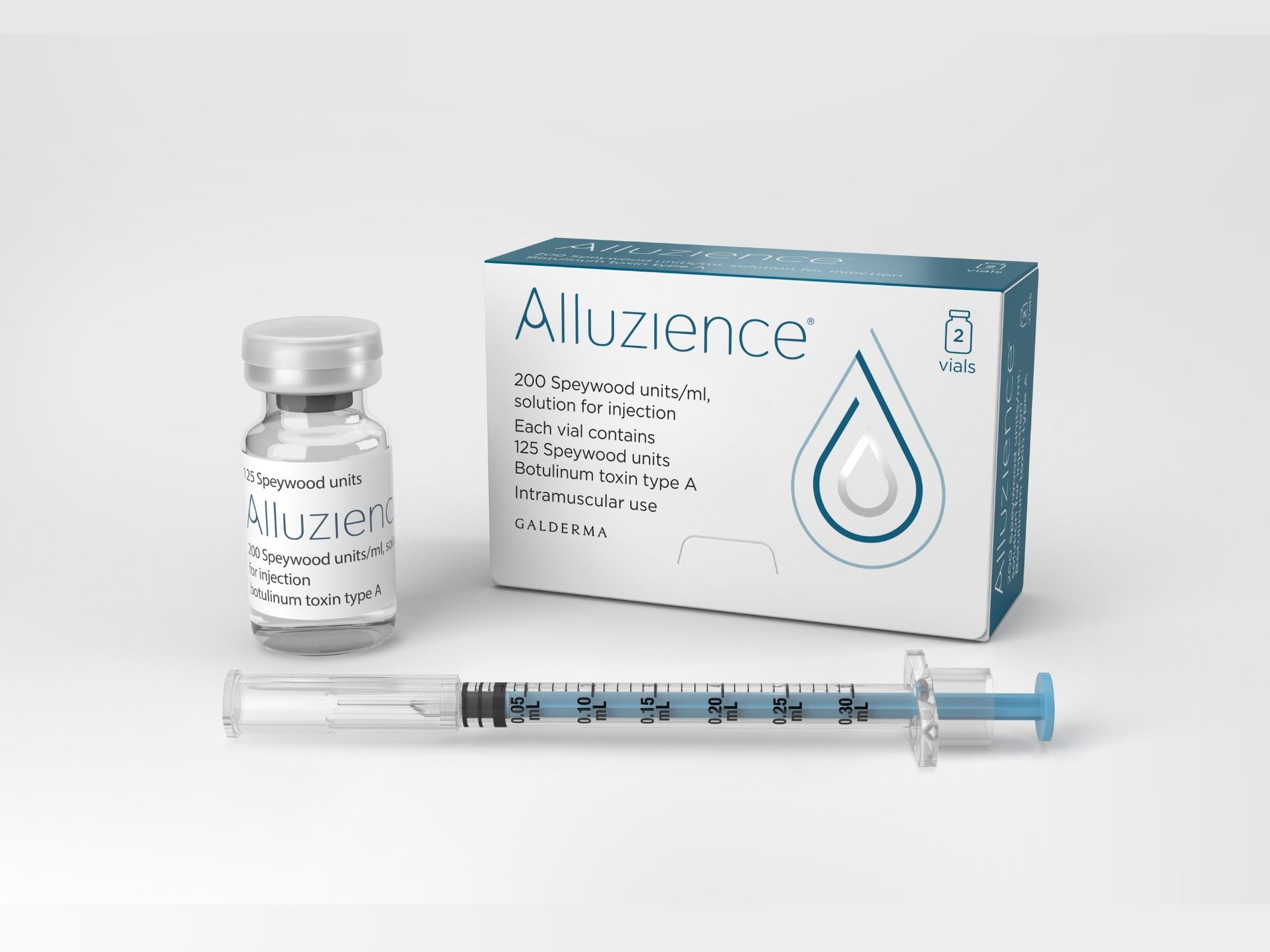
Alluzience (Botulinum Toxin Type A)
The first liquid neuromodulator in Europe, approved for temporary improvement in the appearance of moderate to severe glabellar lines seen at maximum frown in adult patients under 65 years, when the severity of these lines has an important psychological impact on the patient.

Prescribing Information for Azzalure - UK
Prescribing Information for Alluzience - UK
Prescribing Information for Relfydess - UK
UKI-ALA-2500015 March 2025
Adverse events should be reported. For the UK, reporting forms and information can be found at www.mhra.gov.uk/yellowcard or search for MHRA Yellow Card in the Google Play or Apple App Store.
Adverse events should also be reported to Galderma (UK) Ltd. Email: medinfo.uk@galderma.com Tel: +44 (0)300 3035674
*We provide Galderma (U.K.) Limited with certain information on who has purchased their products through this website for consumer service/support and safety purposes. A copy of Galderma’s Privacy Notice explaining how your data will be collected, used, and disclosed by Galderma can be found here.
